|
In the case of ethyl acetate, the reaction is prepared using acids or bases as catalysts and the products: acetic acid and ethanol are produced after a two step mechanism. Ethyl acetate is not soluble in water, but it is soluble in most of the organic solvents such as benzene, acetone, chloroform and toluene.Ĭhemical properties: Ethyl acetate and other esters can be hydrolyzed to the corresponding carboxylic acid and ethanol. Its melting and boiling point are -83.6 ✬ and 77.06 ✬ and its density is 0.894 g mL -1. Physical properties: Ethyl acetate is a colorless, with fruity odor liquid. This esterification reaction is a reversible reaction, so that, it can be added an excess of reagents to improve the yield. The first method is the reaction between ethanol, acetic acid and sulfuric acid, at 60-70 ✬ to promote the esterification of carboxylic acid to ester.ĬH 3COOH + CH 3CH 2OH → CH 3COOC 2H 5 + H 2O Preparation: There are some methods to prepare ethyl acetate. In these drink are added a small quantity of acetic acid which reacts with ethanol to form ethyl acetate, that is responsible for the taste of some old wines. However, it can be found in wine and other alcoholic beverage. Occurrence: Ethyl acetate is not widely present in nature. 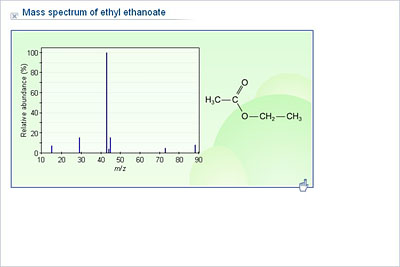
Its chemical structure can be written as below, in the common representations used for organic molecules. 
There is an C sp2 in the carbonyl group, but the other part of molecule has tetrahedral geometry. 
Ethyl acetate is an ester which derive from the replace of hydroxyl group by an ethoxy group in the acetic acid. Ethyl acetate is commonly abbreviated EtOAc. Formula and structure: The ethyl ethanoate chemical formula is CH 3COOCH 2CH 3 and its condensed formula is C 4H 8O 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
